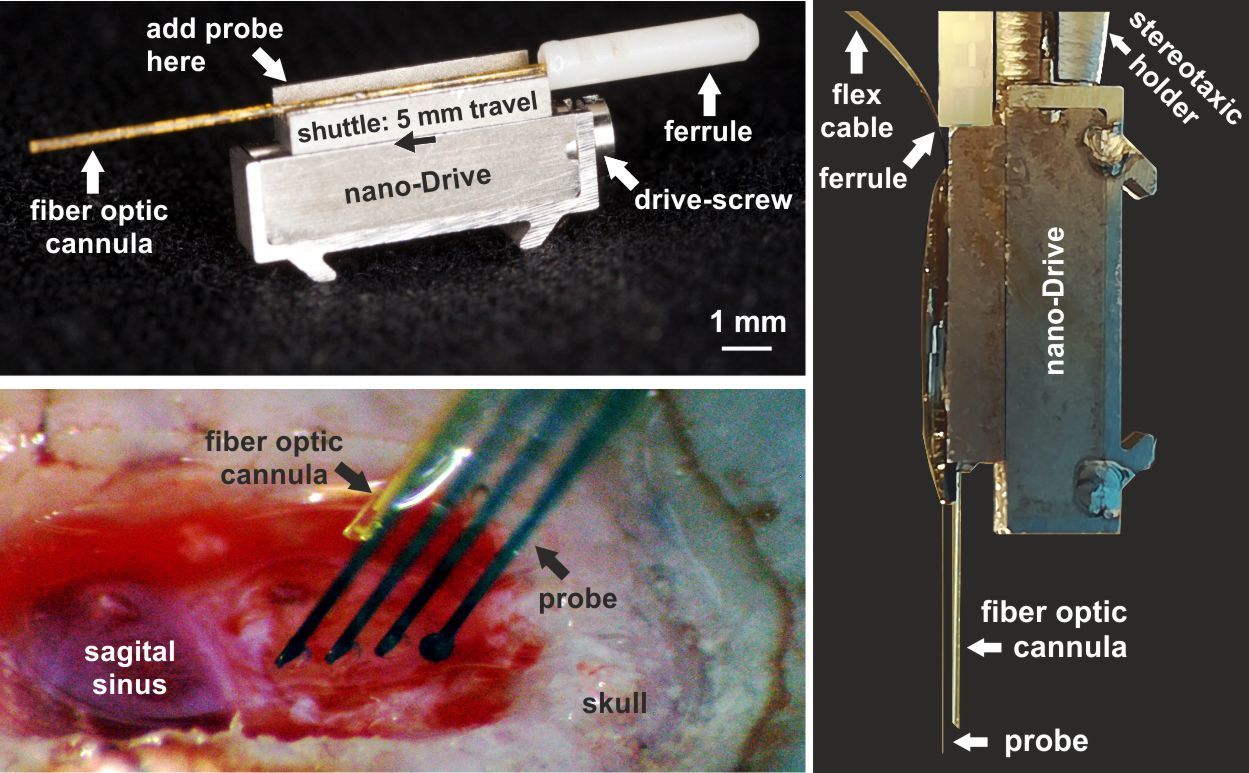
Enjoy artifact-free data with our silicon neural probes during optogenetic stimulation - a major breakthrough for combined electrophysiology with optogenetics.
Photoelectric artifact-free electrophysiology - why it matters...
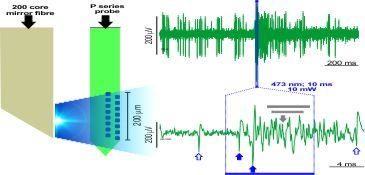
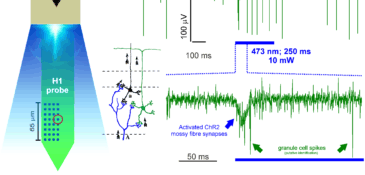
A common problem when combining electrophysiology and optogenetics is that ideally you want your specific target neurons to respond to light but so too will your electrodes since when light strikes a metal electrode it commonly causes a photoelectric artifact due to the Becquerel effect. These artifacts may be large amplitude and long-lasting, resulting in potential data loss and / or contamination of your signal with artifacts that may mimic spikes so single unit recordings during optogenetic stimulation have proved tricky so far....examples of this in the literature can be found in Cardin et al., 2010 and Park et al., 2014; Fig. 4). Our probes are purpose-engineered to be minimally-responsive to light, enabling artifact-free recording of single unit activity during optogenetic stimulation:
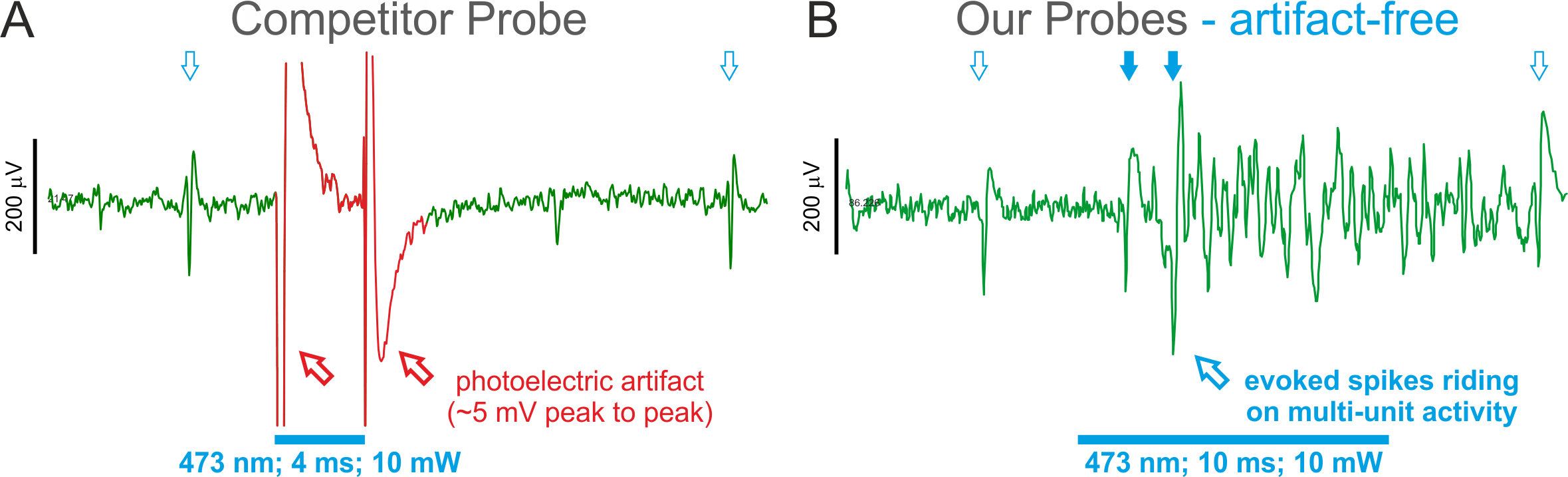
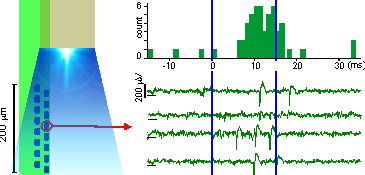
(A) shows a typical photoelectric artifact on a Neuronexus probe evoked by a modest light stimulus delivered to TH::Cre rat VTA neurons expressing ChR2Y - spikes indicated by blue arrows. The artifact is ~25x larger amplitude than the spikes and results in data loss during a period when short latency photo-evoked spikes are most likely to occur. [Data courtesy of Paul Anderson and Mike Cohen, Radboud University, Netherlands] (B) shows evoked photo-evoked activity in the mouse brainstem in a freely-behaving transgenic mouse expressing vGlut2::Cre with ReachR opsin - short-latency evoked spikes are visible in the absence of a photo-electric artifact. [Data courtesy of Ludwig Ruder, Eduardo Arteaga and Silvia Arber, University of Basel, Switzerland]
Combining in vivo optogenetics with electrophysiology is an important and versatile integration with broad relevance since it opens up the possibility to monitor single unit and local network activity during manipulation. This combined approach can be used for photo-identification of single units being recorded, interrogation of the contribution of defined inputs from one region to another to the circuit-level encoding of a stimulus or behavior and moreover for the establishment of closed-loop optogenetic manipulation based on fast-readout of network state / activity.
RELATED LINKS
- Browse our full range of silicon neural probes